

|
|
|
|
What are authoritative sources for scientific
evidence-based and peer-reviewed
information?
The following descriptions briefly explain the difference
between primary, secondary, tertiary, and other sources for
scientific peer-reviewed information.
- Primary Information
Source:
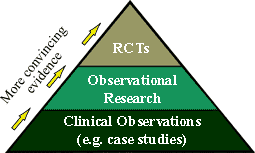 Articles in scientific journals that report the findings
from research studies and experiments are considered
"primary" information sources, because they are
"first hand" accounts of the actual research and the
results, i.e. the original scientific evidence. Papers about
original empirical research must conform to acceptable
scientific standards for conducting and reporting such
research, and must pass peer-review to be published as
articles in a scientific journal. Link
to Examples of Primary Source Scientific Literature. Articles in scientific journals that report the findings
from research studies and experiments are considered
"primary" information sources, because they are
"first hand" accounts of the actual research and the
results, i.e. the original scientific evidence. Papers about
original empirical research must conform to acceptable
scientific standards for conducting and reporting such
research, and must pass peer-review to be published as
articles in a scientific journal. Link
to Examples of Primary Source Scientific Literature. -
- Secondary Information
Source:
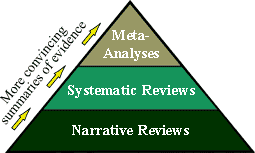 The various original scientific studies described above
generate a wealth of "primary evidence" that
periodically needs to be gathered, organized, and assessed by
the clinical and scientific communities to give us a sense of
the current "state of the science" on a given topic.
These types of research "Review" articles are
considered a secondary source because they are
"second-hand" accounts which integrate the
scientific evidence from a number of original research
studies. However, such scientific "Review" articles
must also conform to scientifically acceptable methodologic
standards for gathering, synthesizing, and reporting the available
evidence, and also must be peer-reviewed before being
published in a reputable scientific journal. Link
to Examples of Secondary Source Scientific Literature. The various original scientific studies described above
generate a wealth of "primary evidence" that
periodically needs to be gathered, organized, and assessed by
the clinical and scientific communities to give us a sense of
the current "state of the science" on a given topic.
These types of research "Review" articles are
considered a secondary source because they are
"second-hand" accounts which integrate the
scientific evidence from a number of original research
studies. However, such scientific "Review" articles
must also conform to scientifically acceptable methodologic
standards for gathering, synthesizing, and reporting the available
evidence, and also must be peer-reviewed before being
published in a reputable scientific journal. Link
to Examples of Secondary Source Scientific Literature.-
- Tertiary Information
Sources:
- Textbooks offer an example of a usually reliable source of
information, that may or may not be peer-reviewed. The more
accurate and reliable textbooks will contain information that
is drawn only from the evidence from primary and secondary
source articles that have been published in the peer-reviewed
scientific literature, and that conform to scientific
standards for summarizing and interpreting that
information. For instance, the author(s) of an accurate
and reliable textbook will appropriately and objectively
summarize all information available on a topic, and not just
"pick and choose" certain evidence or interpret the
selected evidence in a biased manner.
-
- In accredited institutions, textbooks
are reviewed and approved by the faculty of the institution,
which provides another level of "peer-review" of the
information in the textbook. Textbooks in wide use by a number
of accredited institutions may give a further indication of
the extent to which educators, clinicians, and scientists
consider the book a credible and legitimate source of accurate
and complete information.
-
- Other authoritative
peer-reviewed sources:
- Information from other sources may be considered
"peer-reviewed", in that the information is
reviewed, approved, and regularly updated by a group of
recognized authorities such as health profession
organizations, accredited institutions and universities, and
governmental entities. Such organizations may produce and
disseminate monographs and reports that offer synopses of
"current evidence" for health care professionals. An
example of such a "synopsis of current evidence" is
the report from the Institute of Medicine "Immunization
Safety Review: Multiple Immunizations and Immune
Dysfunction", available via the IOM web links provided
below.
-
- Websites maintained or sanctioned by such recognized
authorities are also a source for brief information summaries
such as FAQ pages (Frequently Asked Questions) or "Fact
Sheets" that have been compiled by clinical experts and
scientists in public health and immunology. These FAQ/Fact
Sheets synthesize complex scientific evidence and present it
in a way that can be more easily read and understood by
patients and the "typical" health care consumer.
Such FAQ/Fact summaries of scientific evidence-based
information can usually be downloaded directly from the
Internet.
-
- Examples of online FAQ/Fact Sheets on vaccination include the
following links to the World Health Organization, the U.S. Centers for Disease
Control and Prevention, and the Institute of Medicine. Also included are links to useful guides that can
help health care consumers "sort through" and
determine whether various Internet-based information comes
from a credible, authoritative source.
-
- These links are
provided only as examples of the many sources and types of
vaccination information available to consumers on the Internet.
The authors of the Immunization Information Resource website
do not take responsibility for the content of other websites,
nor for evaluating or endorsing that content.
|
|
|
|
Links:
Institute of Medicine
The
Institute of Medicine (IOM) is a branch of the National Academy of
Sciences providing health-related information to the government,
corporate sector, professions and the public. http://www.iom.edu/imsafety
World Health Organization
The World Health
Organization (WHO) is an international organization concerned with
the attainment of health.
http://www.who.int/vaccines-diseases
Centers for Disease Control and Prevention (CDC)
The Centers for Disease Control and Prevention (CDC) is the U.S.
Federal agency charged with protecting the health and safety of
the general population.
http://www.cdc.gov/od/nvpo
Hopkins Health
Faculty from Johns Hopkins Schools of Medicine, Public Health or
Nursing take responsibility for the accuracy of all information
provided in this database of health-related documents, with no
editorial content from faculty with any conflict of interest.
http://www.vaccinesafety.edu
Health Summit Working Group: Criteria for Assessing the Quality
of Health Information on the Internet
http://hitiweb.mitretek.org/docs/policy.html#eval
CDC: 10 Tips on Evaluating Immunization Information on the
Internet:
http://www.cdc.gov/od/nvpo/tips.htm
|
|
|

[ Top of Page ] [ Up ]
[ Home
]
|
|
Last updated on: 09/05/2002
|
|