| |
|
Examples
of Primary Source and Secondary Source
Evidence-Based and Peer-Reviewed Scientific Literature
|
 |
|
|
|
|
|
Criteria for Scientifically
Establishing Cause-and-Effect:
Scientific research mainly seeks to answer questions of cause
and effect. In health care, much of the clinical and epidemiologic
research is concerned with what causes a health condition such as
disease, pain, or dysfunction, and therefore what might be done to
prevent the health condition from occurring in the first place.
Or
in cases where a health condition is already present, what
interventions can help to return a patient to a more healthy state
(i.e. to assist in causing healing to occur), or at the very least
slow down or stop a condition from progressing to a worse state.
Scientific inquiry is based on "common
sense" principles for determining cause-and-effect.
The following Table 1 outlines some of the "Criteria for Causality" that must be met to build a
convincing "body of scientific evidence" of a
cause-and-effect relationship. Does the hypothesized cause of the
condition (e.g. exposure of the patient to a "risk
factor") occur before the observed effect (e.g. the onset of
illness). Are there other possible factors that have also
occurred, and that could have contributed to causing the illness?
Does the established scientific knowledge-base or evidence from
other previous research convincingly support the hypothesized cause-and-effect
relationship?
|
|
|
Table 1: Examples of Criteria for Establishing Causality
(Cause-and-Effect) in Clinical Research
 | Time-Order
> Does the hypothesized
cause precede
the effect?
> Can the time-order of cause-and-effect
be determined from the Observational or
Quasi-Experimental Research Study, or has time-order
been "controlled" in the Experimental Study?
|
 | Specificity of Cause-and-Effect Association
> Are there other possible causes for
the observed effect?
> Have other possible causative factors been
considered or "controlled" in the Research
Study?
|
 | Consistency and Scientific
Plausibility
> Consistent results across numerous and varied
Clinical or Epidemiologic Research Studies will provide stronger
cumulative evidence in support of a postulated cause-and-effect
relationship.
> Do the findings from the Clinical and Epidemiological
Research (Studies of cause-and-effect at the
"person-level") "make sense"
relative to what we know from Basic Science Research
(e.g. Studies of cause-and-effect at the cellular
level)?
|
|
|
|
|

|
|
Primary Source Evidence:
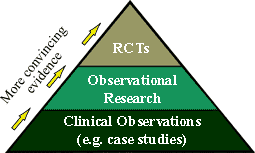 The
second table (Table 2, below) titled "Continuum of Study
Designs and their Causal Inference" demonstrates the
hierarchy of the different types of research studies. This
hierarchy is based on the ability of the research study design to
satisfy the "Criteria for Causality" (some of which were
identified in Table 1 above). The research examples at the bottom of the table (e.g.
case studies) are good for beginning the process of developing
cause-and-effect research hypotheses, but such studies can only
provide very weak evidence since they don't meet many of the
Criteria for establishing Causality. Moving from the bottom to the
top of
Table 2, the rigor of the research studies is
increased, more of the causal criteria are met, and a stronger
case may be made for drawing conclusions (inferences) about
cause-and-effect. At the top of Table 2 is the experimental
randomized controlled trial (RCT), which is the strongest study design
for meeting the causal criteria, and therefore is the "gold
standard" of scientific evidence. The
second table (Table 2, below) titled "Continuum of Study
Designs and their Causal Inference" demonstrates the
hierarchy of the different types of research studies. This
hierarchy is based on the ability of the research study design to
satisfy the "Criteria for Causality" (some of which were
identified in Table 1 above). The research examples at the bottom of the table (e.g.
case studies) are good for beginning the process of developing
cause-and-effect research hypotheses, but such studies can only
provide very weak evidence since they don't meet many of the
Criteria for establishing Causality. Moving from the bottom to the
top of
Table 2, the rigor of the research studies is
increased, more of the causal criteria are met, and a stronger
case may be made for drawing conclusions (inferences) about
cause-and-effect. At the top of Table 2 is the experimental
randomized controlled trial (RCT), which is the strongest study design
for meeting the causal criteria, and therefore is the "gold
standard" of scientific evidence.
|
|
|
|
|
|
|
Table 2: Continuum of Examples of Study Designs and their Causal Inference |
|
| |
Study Designation |
(1) |
|
(2) |
|
(3) |
|
Inference |
|
| |
Clinical randomized trials |
|
|
|
|
x |
|
Firm |
More
convincing
evidence
of cause-
and-effect |
| |
Community randomized trials |
|
|
|
|
x |
|
Firm |
| |
Cohort |
|
|
x |
|
|
|
Moderately
Firm |
| |
Case-control |
|
|
x |
|
|
|
More
Suggestive |
| |
Cross-sectional |
|
|
x |
|
|
|
Suggestive |
| |
Time series |
x |
|
x |
|
|
|
Suggestive |
| |
Case series |
x |
|
|
|
|
|
Speculative |
| |
Case study |
x |
|
|
|
|
|
Speculative |
(1) Clinical Observations, (2)
Observational and "Quasi-Experimental" Research,
(3) Experimental Research (e.g. Randomized
Controlled Trials - RCTs) | |
|
|
|
|
|
|
Following are brief descriptions and examples of
some of the different types of "Primary Source" research
studies listed in Table 2.
Case Report, Case Study, Case Series: In these
"clinical observations", a clinician reports
"out of the ordinary" findings from their clinical
experience with a patient (a "case") or a number of
similar patients (a "series"). These reports of
patients' clinical histories are typically used to notify the
clinical scientific community of a new and noteworthy aspect of a
condition, clinical intervention, or adverse reaction.
Cross-Sectional studies are frequently surveys conducted
at one point in time, for instance to determine the prevalence of
a certain health condition among a defined population.
Case-Control studies identify a group of
"cases" (those with a condition) and a group of
"controls" (those without the condition). The
researcher looks back in time and compares the prior history of
the "cases" to the "controls", to identify
possible causative risk factors that preceded the development of
the condition. When enough scientific evidence has been
accumulated through case-control studies to support a possible
cause-and-effect finding, then it justifies spending the time and
resources to fund and conduct more rigorous, more difficult, and
more expensive Cohort studies. A Cohort study starts by first identifying individuals
with a suspected risk factor and comparing them to individuals
without the risk factor. This cohort is followed forward over a
period of time to determine whether the presence of the suspected
causative risk factor is associated with the development of the
condition (the effect).
Randomized Controlled Trials (RCTs) are experimental
studies in which the researcher can control various important
factors, and thereby systematically and convincingly "rule
out" other possible explanations for an observed apparent cause-and-effect relationship. This is the strongest and most
rigorous study design for meeting the "Criteria for
Causality", which is why it is considered the "Gold
Standard" of clinical research.
|
|
|

|
|
Secondary Source Summaries of Evidence:
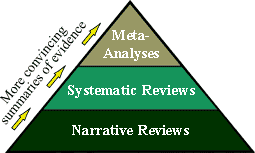 The
"primary evidence" from the research studies described
above is periodically "Reviewed" by gathering all of the
evidence available on a given topic, and comparing and contrasting
the various research studies and the results from those
studies. Review articles follow acceptable scientific methods
which again make use of the common-sense "Criteria for
Causality" to compare and contrast the strength of the
different studies for supporting (or refuting) a hypothesized
cause-and-effect relationship. Generally, Review articles fall
into one of 3 broad categories in order of increasing scientific
rigor: the narrative review, the systematic review, and
meta-analyses, which are briefly described below. The
"primary evidence" from the research studies described
above is periodically "Reviewed" by gathering all of the
evidence available on a given topic, and comparing and contrasting
the various research studies and the results from those
studies. Review articles follow acceptable scientific methods
which again make use of the common-sense "Criteria for
Causality" to compare and contrast the strength of the
different studies for supporting (or refuting) a hypothesized
cause-and-effect relationship. Generally, Review articles fall
into one of 3 broad categories in order of increasing scientific
rigor: the narrative review, the systematic review, and
meta-analyses, which are briefly described below.
Narrative Reviews are essentially "essays" wherein
the researcher/author focuses on a particular clinical topic, and
presents in their opinion what they feel are some of the
highlights worth noting from the scientific literature they review. Narrative Reviews are the weakest of the
"Secondary Source" research literature, since the
methods and the reporting are somewhat subjective, and therefore
may be more prone to bias. Articles that report original empirical
research studies (described as "Primary Source" evidence
above) typically also contain good "narrative overviews"
of the "existing evidence" as an introduction and
background to the article, to explain and justify why the original
research study was conducted.
Systematic Reviews are conducted according to scientific
methodologies that are explicit, explicitly stated, and
reproducible. For instance, the methods section of a systematic
review will describe the literature search in enough detail (e.g.
what databases were searched, for what years, search terms
employed), that the search strategy could be replicated by another
researcher and produce essentially the same yield of literature.
The "primary evidence" articles are then systematically
and explicitly compared and contrasted along a number of important
dimensions of research designs that reflect the "Criteria for
Causality", which makes for a stronger and more useful
"review of the resulting evidence" than do simple
narrative reviews. This "attention to detail" in
conducting and reporting the systematic review, helps the readers
of the review article (i.e. the clinical and scientific
communities) to determine how strong the accumulated evidence
appears to be, without relying solely on the opinion of the
author.
Meta-Analyses take the systematic review to the highest and
most rigorous level of integrating "primary evidence"
research, by statistically synthesizing the quantitative results
across similar studies. Essentially, meta-analyses pool the data and
results from a number of original research studies, with the
intent of producing a more precise summary of the existing
evidence for (or against) a cause-and-effect relationship.
For example, a meta-analysis may collect results from a number of
epidemiologic studies, and statistically combine the data to
produce a more precise overall "risk estimate" of the
relationship between the causative risk factor and the effect
(the disease). |
|
|
|

[ Top of Page ] [ Up
] [ Home
]
|
|